 It is important to note that for both transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. The electrons of incomplete d-orbitals may also be considered as valence electrons in some inner transition metals. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry-and they do. Similarly, for inner transition elements (or f-block elements), the valence electrons can range from 3 to 16 due to the contribution of both the f-subshell and s-subshell electrons. They all have a similar electron configuration in their valence shells: a single s electron. In some cases, the electrons of incomplete d-orbitals are also considered as valence electrons. This is because the d-subshell of transition elements and f-subshell of inner transition elements are incompletely filled and are very close to the outer s-subshell.Īs a result, the electrons of both the d-subshell and s-subshell in transition elements and f-subshell and s-subshell in inner transition elements can behave like valence electrons.įor transition elements, the valence electrons can range from 3 to 12 due to the contribution of both the d-subshell and s-subshell electrons. Counting valence electrons for main group elements. Valence electrons of transition elements and inner transition elements are more complicated to determine than those of main group elements. Valence electrons of transition and inner-transition elements These valence electrons are responsible for the elements’ unique chemical properties and reactivity. The number of valence electrons in these elements is equal to their group number.įor example, elements in group 1, such as lithium and sodium, have one valence electron, while elements in group 2, such as magnesium and calcium, have two valence electrons.Įlements in group 13, such as boron and aluminum, have three valence electrons, and so on. The main group elements, also known as the representative elements, are located in groups 1, 2, and 13-18 of the periodic table. The third lithium electron (this is the valence electron). Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. electrons in the n 1 state, one with quantum numbers (1,0, 0, 1/2), the other with (1, 0, 0, 1/2). The number of valence electrons an atom has determines its position in the periodic table and its ability to form chemical bonds. Thus the inner transition elements have valence electrons ranging from 3 to 16 (See above image of periodic table). These electrons are responsible for the chemical properties of an element, such as its reactivity and the types of chemical bonds it can form with other atoms. The lithium atoms have only one valence electron, which is very easy to lose. 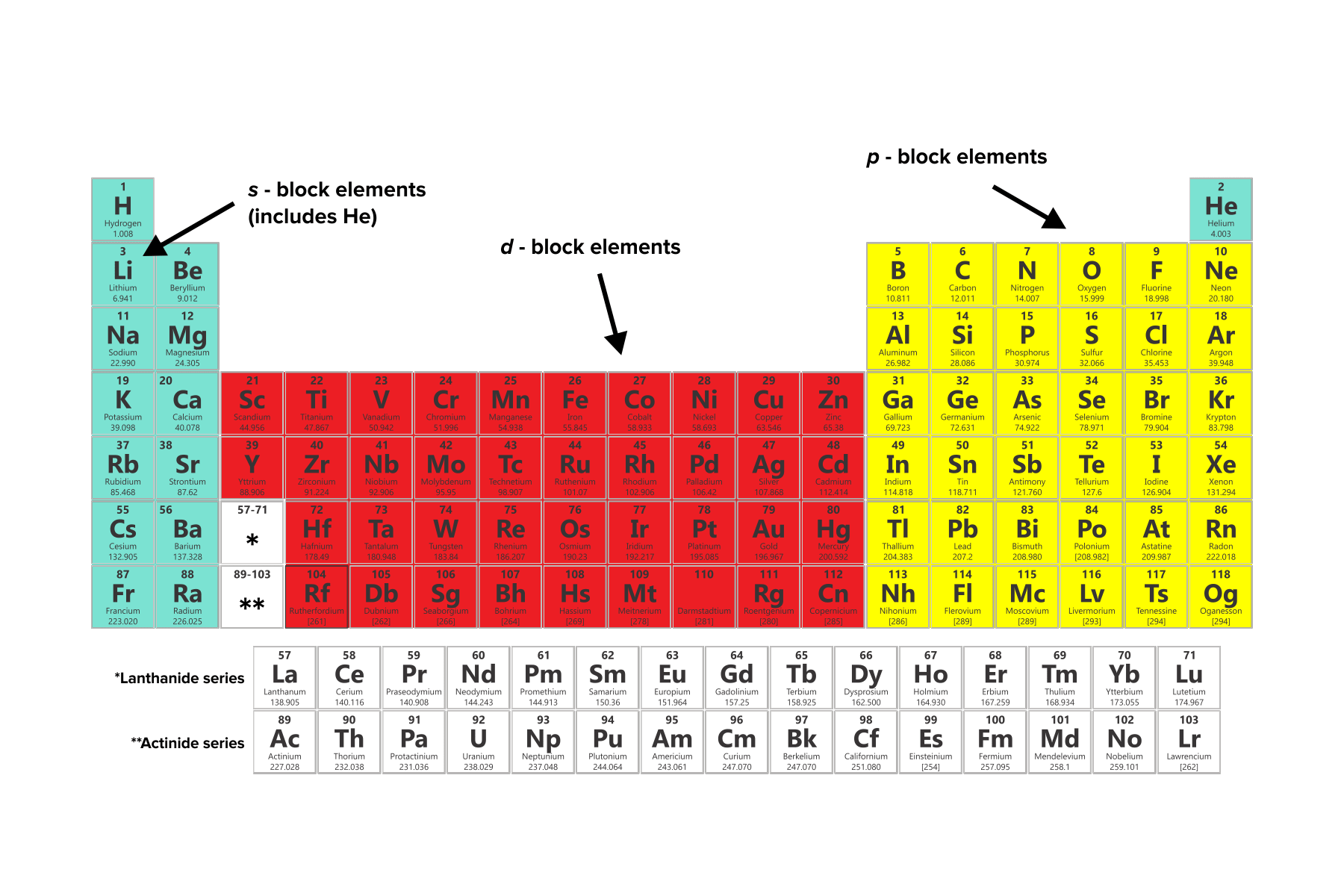
Valence electrons are the electrons located in the outermost shell or energy level of an atom that are involved in chemical bonding.įor example, magnesium has 2 electrons in its outermost orbit, hence it has 2 valence electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
